what blood pressure ill is equivalent to bystolic
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Nebilet, Bystolic, others |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a608029 |
| License data |
|
| Pregnancy category |
|
| Routes of administration | By mouth |
| ATC code |
|
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Protein bounden | 98% |
| Metabolism | Liver (CYP2D6-mediated) |
| Elimination half-life | 10 hours |
| Excretion | Kidney and fecal |
| Identifiers | |
| CAS Number |
|
| PubChem CID |
|
| IUPHAR/BPS |
|
| DrugBank |
|
| ChemSpider |
|
| UNII |
|
| KEGG |
|
| ChEMBL |
|
| CompTox Dashboard (EPA) |
|
| Chemic and concrete data | |
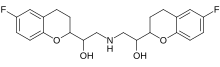
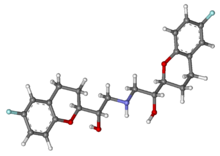
| Formula | C 22 H 25 F 2 N O iv |
| Molar mass | 405.442 grand·mol−1 |
| 3D model (JSmol) |
|
| SMILES
| |
| InChI
| |
| | |
Nebivolol is a beta blocker used to care for high blood pressure and heart failure.[i] Equally with other β-blockers, it is generally a less preferred treatment for loftier blood pressure.[two] Information technology may exist used by itself or with other blood pressure medication.[two] It is taken by mouth.[2]
Mutual side effects include dizziness, feeling tired, nausea, and headaches.[two] Serious side effects may include heart failure and bronchospasm.[ii] Its employ in pregnancy and breastfeeding is not recommended.[ane] [iii] It works past blocking β1-adrenergic receptors in the center and dilating blood vessels.[2] [4]
Nebivolol was patented in 1983 and came into medical employ in 1997.[5] It is available as a generic medication in the Great britain.[one] In 2019, it was the 191st well-nigh commonly prescribed medication in the U.s., with more than than threemillion prescriptions.[six] [7]
Medical uses [edit]
Information technology is used to treat loftier blood pressure and heart failure.[1] Nebivolol is used in the treatment of angina, to decrease the heart rate and contractile force. This is relevant in patients who demand to decrease the oxygen demand of the heart so that the blood supplied from stenosed or constricted arteries be adequate.
ACE inhibitors, angiotensin II receptor antagonists, calcium-channel blockers, and thiazide diuretics are generally preferred over beta blockers for the treatment of primary hypertension in the absence of co-morbidities.[viii] [9] [x] [xi]
Contraindications [edit]
- Severe bradycardia
- Heart block greater than first caste
- Patients with cardiogenic daze
- Decompensated cardiac failure
- Sick sinus syndrome (unless a permanent pacemaker is in place)
- Patients with severe hepatic impairment (Kid-Pugh course B)
- Patients who are hypersensitive to any component of this product.
Adverse reactions [edit]
- Headache
- Paresthesia
- Dizziness
- Deadening middle rate
Pharmacology and biochemistry [edit]
β1-selectivity [edit]
Beta blockers help patients with cardiovascular affliction by blocking β receptors, while many of the side-furnishings of these medications are caused by their blockade of β2 receptors.[12] For this reason, beta blockers that selectively block β1 adrenergic receptors (termed cardioselective or β1-selective beta blockers) produce fewer adverse effects (for case, bronchoconstriction) than those drugs that not-selectively block both β1 and β2 receptors.
In a laboratory experiment conducted on biopsied eye tissue, nebivolol proved to be the most β1-selective of the β-blockers tested, being approximately 3.v times more β1-selective than bisoprolol.[xiii] Yet, the drug'due south receptor selectivity in humans is more complex and depends on the drug dose and the genetic profile of the patient taking the medication.[14] The drug is highly cardioselective at v mg.[15] In addition, at doses above x mg, nebivolol loses its cardioselectivity and blocks both β1 and β2 receptors.[14] (While the recommended starting dose of nebivolol is v mg, sufficient command of blood force per unit area may require doses up to twoscore mg).[14] Furthermore, nebivolol is besides non cardioselective when taken by patients with a genetic makeup that makes them "poor metabolizers" of nebivolol (and other drugs) or with CYP2D6 inhibitors.[14] Every bit many as i in 10 Caucasian people and even more than black people are poor CYP2D6 metabolizers and therefore might benefit less from nebivolol's cardioselectivity although currently there are no straight comparable studies.[ commendation needed ]
Nebivolol[sixteen] while selectively blocking beta(i) receptor acts as a beta(3)-agonist. βiii receptors are constitute in the gallbladder, urinary bladder, and in brown adipose tissue. Their role in gallbladder physiology is unknown, only they are thought to play a part in lipolysis and thermogenesis in brown fat. In the urinary bladder it is thought to cause relaxation of the bladder and prevention of urination.[17] [18] [19]
Due to enzymatic inhibition, fluvoxamine increases the exposure to nebivolol and its active hydroxylated metabolite (iv-OH-nebivolol) in healthy volunteers.[twenty]
Vasodilator action [edit]
Nebivolol is unique as a beta-blocker.[21] Unlike carvedilol, it has a nitric oxide (NO)-potentiating, vasodilatory effect via stimulation of β3 receptors.[22] [23] [24] Along with labetalol, celiprolol and carvedilol, it is one of four beta blockers to cause dilation of claret vessels in improver to furnishings on the heart.[24]
Antihypertensive result [edit]
Nebivolol lowers blood pressure level (BP) by reducing peripheral vascular resistance, and significantly increases stroke volume with preservation of cardiac output.[25] The net hemodynamic upshot of nebivolol is the outcome of a residual between the depressant furnishings of beta-blockade and an action that maintains cardiac output.[26] Antihypertensive responses were significantly higher with nebivolol than with placebo in trials enrolling patient groups considered representative of the U.S. hypertensive population, in black people, and in those receiving concurrent treatment with other antihypertensive drugs.[27]
Pharmacology of side-effects [edit]
Several studies have suggested that nebivolol has reduced typical beta-blocker-related side effects, such every bit fatigue, clinical depression, bradycardia, or impotence.[28] [29] [thirty] However, according to the FDA[31]
-
Bystolic is associated with a number of serious risks. Bystolic is contraindicated in patients with severe bradycardia, heart block greater than first degree, cardiogenic shock, decompensated cardiac failure, sick sinus syndrome (unless a permanent pacemaker is in place), severe hepatic impairment (Child-Pugh > B) and in patients who are hypersensitive to whatsoever component of the product. Bystolic therapy is besides associated with warnings regarding abrupt abeyance of therapy, cardiac failure, angina and acute myocardial infarction, bronchospastic diseases, anesthesia and major surgery, diabetes and hypoglycemia, thyrotoxicosis, peripheral vascular disease, non-dihydropyridine calcium channel blockers use, as well every bit precautions regarding employ with CYP2D6 inhibitors, impaired renal and hepatic role, and anaphylactic reactions. Finally, Bystolic is associated with other risks as described in the Adverse Reactions section of its PI. For example, a number of treatment-emergent adverse events with an incidence greater than or equal to 1 percent in Bystolic-treated patients and at a higher frequency than placebo-treated patients were identified in clinical studies, including headache, fatigue, and dizziness.
FDA alert letter nearly advertising claims [edit]
In belatedly August 2008, the FDA issued a Alert Letter to Wood Laboratories citing exaggerated and misleading claims in their launch journal advertizement, in particular over claims of superiority and novelty of action.[31]
History [edit]
Mylan Laboratories licensed the U.S. and Canadian rights to nebivolol from Janssen Pharmaceutica N.V. in 2001. Nebivolol is already registered and successfully marketed in more than than 50 countries, including the United States where it is marketed under the brand proper name Bystolic from Mylan Laboratories and Forest Laboratories. Nebivolol is manufactured by Forest Laboratories.
In India, nebivolol is bachelor as Nebula (Zydus Healthcare Ltd), Nebizok (Eris life-sciences), Nebicip (Cipla ltd), Nebilong (Micro Labs), Nebistar (Lupin ltd), Nebicard (Torrent), Nubeta (Abbott Healthcare Pvt Ltd – India), and Nodon (Cadila Pharmaceuticals). In Greece and Italy, nebivolol is marketed by Menarini as Lobivon. In the Middle East, Russia and in Australia, it is marketed under the proper name Nebilet and in Pakistan information technology is marketed by The Searle Company Limited every bit Byscard.
References [edit]
- ^ a b c d British national formulary : BNF 76 (76 ed.). Pharmaceutical Press. 2018. p. 154. ISBN9780857113382.
- ^ a b c d e f "Nebivolol Hydrochloride Monograph for Professionals". Drugs.com. American Guild of Health-Organisation Pharmacists. Retrieved 3 March 2019.
- ^ "Nebivolol Pregnancy and Breastfeeding Warnings". Drugs.com . Retrieved 3 March 2019.
- ^ de Boer RA, Voors AA, van Veldhuisen DJ (July 2007). "Nebivolol: third-generation beta-blockade". Practiced Opinion on Pharmacotherapy. eight (10): 1539–50. doi:10.1517/14656566.8.10.1539. PMID 17661735. S2CID 24186687.
- ^ Fischer J, Ganellin CR (2006). Analogue-based Drug Discovery. John Wiley & Sons. p. 462. ISBN9783527607495.
- ^ "The Elevation 300 of 2019". ClinCalc . Retrieved 16 October 2021.
- ^ "Nebivolol - Drug Usage Statistics". ClinCalc . Retrieved xvi October 2021.
- ^ Reboussin DM, Allen NB, Griswold ME, Guallar E, Hong Y, Lackland DT, et al. (June 2018). "Systematic Review for the 2017 ACC/AHA/AAPA/ABC/ACPM/AGS/APhA/ASH/ASPC/NMA/PCNA Guideline for the Prevention, Detection, Evaluation, and Management of Loftier Blood Force per unit area in Adults: A Study of the American College of Cardiology/American Middle Association Task Strength on Clinical Practice Guidelines". Hypertension. 71 (6): e116–e135. doi:10.1161/HYP.0000000000000067. PMID 29133355. S2CID 34462045.
- ^ Fretheim A, Odgaard-Jensen J, Brørs O, Madsen S, Njølstad I, Norheim OF, et al. (April 2012). "Comparative effectiveness of antihypertensive medication for primary prevention of cardiovascular disease: systematic review and multiple treatments meta-analysis". BMC Medicine. 10 (1): 33. doi:10.1186/1741-7015-10-33. PMC3354999. PMID 22480336.
- ^ Seleme VB, Marques GL, Mendes AE, Rotta I, Pereira Thousand, Júnior EL, da Cunha CL (March 2021). "Nebivolol for the Handling of Essential Systemic Arterial Hypertension: A Systematic Review and Meta-Analysis". American Journal of Cardiovascular Drugs. 21 (ii): 165–180. doi:10.1007/s40256-020-00422-0. PMID 32710438. S2CID 220732879.
- ^ National Heart Foundation of Australia. National Blood Pressure and Vascular DiseaseAdvisory Commission (2016). Guideline for the diagnosis and direction of hypertension in adults. Melbourne, Vic. ISBN978-ane-74345-110-6. OCLC 970693159.
- ^ Tafreshi MJ, Weinacker AB (August 1999). "Beta-adrenergic-blocking agents in bronchospastic diseases: a therapeutic dilemma". Pharmacotherapy. 19 (8): 974–eight. doi:10.1592/phco.19.11.974.31575. PMID 10453968. S2CID 32202416.
- ^ Bundkirchen A, Brixius K, Bölck B, Nguyen Q, Schwinger RH (January 2003). "Beta one-adrenoceptor selectivity of nebivolol and bisoprolol. A comparing of [3H]CGP 12.177 and [125I]iodocyanopindolol binding studies". European Journal of Pharmacology. 460 (1): 19–26. doi:10.1016/S0014-2999(02)02875-3. PMID 12535855.
- ^ a b c d "Prescribing information for Bystolic" (PDF) . Retrieved 11 June 2009.
- ^ Nuttall SL, Routledge HC, Kendall MJ (June 2003). "A comparing of the beta1-selectivity of three beta1-selective beta-blockers". Periodical of Clinical Pharmacy and Therapeutics. 28 (iii): 179–86. doi:x.1046/j.1365-2710.2003.00477.x. PMID 12795776. S2CID 58760796.
- ^ Rozec B, Erfanian M, Laurent K, Trochu JN, Gauthier C (April 2009). "Nebivolol, a vasodilating selective beta(ane)-blocker, is a beta(3)-adrenoceptor agonist in the nonfailing transplanted human middle". Periodical of the American College of Cardiology. 53 (17): 1532–8. doi:10.1016/j.jacc.2008.11.057. PMID 19389564.
- ^ Sawa M, Harada H (2006). "Recent developments in the design of orally bioavailable beta3-adrenergic receptor agonists". Current Medicinal Chemical science. 13 (one): 25–37. doi:10.2174/092986706775198006. PMID 16457637.
- ^ Ferrer-Lorente R, Cabot C, Fernández-López JA, Alemany 1000 (September 2005). "Combined effects of oleoyl-estrone and a beta3-adrenergic agonist (CL316,243) on lipid stores of diet-induced overweight male Wistar rats". Life Sciences. 77 (sixteen): 2051–8. doi:10.1016/j.lfs.2005.04.008. PMID 15935402.
- ^ Rang HP, Dale MM, Ritter JM, Moore PK (2003). Pharmacology (fifth ed.). Edinburgh: Churchill Livingstone. p. 163. ISBN0-443-07145-4.
- ^ Gheldiu AM, Vlase 50, Popa A, Briciu C, Muntean D, Bocsan C, et al. (2017). "Investigation of a Potential Pharmacokinetic Interaction Between Nebivolol and Fluvoxamine in Good for you Volunteers". Journal of Pharmacy & Pharmaceutical Sciences. 20: 68–80. doi:x.18433/J3B61H. PMID 28459657.
- ^ Agabiti Rosei E, Rizzoni D (2007). "Metabolic contour of nebivolol, a beta-adrenoceptor antagonist with unique characteristics". Drugs. 67 (8): 1097–107. doi:10.2165/00003495-200767080-00001. PMID 17521213. S2CID 25807936.
- ^ Karimi Galougahi K, Liu CC, Garcia A, Gentile C, Fry NA, Hamilton EJ, et al. (Feb 2016). "β3 Adrenergic Stimulation Restores Nitric Oxide/Redox Balance and Enhances Endothelial Function in Hyperglycemia". Journal of the American Heart Clan. five (2): e002824. doi:10.1161/JAHA.115.002824. PMC4802476. PMID 26896479.
- ^ Weiss R (2006). "Nebivolol: a novel beta-blocker with nitric oxide-induced vasodilatation". Vascular Health and Run a risk Management. 2 (iii): 303–8. doi:10.2147/vhrm.2006.two.iii.303. PMC1993984. PMID 17326335.
- ^ a b Bakris Yard (May 2009). "An in-depth analysis of vasodilation in the management of hypertension: focus on adrenergic occludent". Journal of Cardiovascular Pharmacology. 53 (5): 379–87. doi:10.1097/FJC.0b013e31819fd501. PMID 19454898. S2CID 205598744.
- ^ Kamp O, Sieswerda GT, Visser CA (August 2003). "Comparing of furnishings on systolic and diastolic left ventricular role of nebivolol versus atenolol in patients with uncomplicated essential hypertension". The American Periodical of Cardiology. 92 (3): 344–eight. doi:x.1016/S0002-9149(03)00645-3. PMID 12888152.
- ^ Gielen W, Cleophas TJ, Agrawal R (August 2006). "Nebivolol: a review of its clinical and pharmacological characteristics". International Journal of Clinical Pharmacology and Therapeutics. 44 (viii): 344–57. doi:ten.5414/cpp44344. PMID 16961165.
- ^ Baldwin CM, Keam SJ (2009). "Nebivolol: in the treatment of hypertension in the Usa". American Journal of Cardiovascular Drugs: Drugs, Devices, and Other Interventions. 9 (iv): 253–threescore. doi:x.2165/1120274-000000000-00000. PMID 19655820.
- ^ Pessina AC (December 2001). "Metabolic effects and safety contour of nebivolol". Journal of Cardiovascular Pharmacology. 38. 38 (Suppl 3): S33-five. doi:10.1097/00005344-200112003-00006. PMID 11811391. S2CID 20786564.
- ^ Weber MA (December 2005). "The role of the new beta-blockers in treating cardiovascular disease". American Periodical of Hypertension. 18 (12 Pt ii): 169S–176S. doi:10.1016/j.amjhyper.2005.09.009. PMID 16373195.
- ^ Poirier Fifty, Cléroux J, Nadeau A, Lacourcière Y (Baronial 2001). "Effects of nebivolol and atenolol on insulin sensitivity and haemodynamics in hypertensive patients". Periodical of Hypertension. xix (8): 1429–35. doi:10.1097/00004872-200108000-00011. PMID 11518851. S2CID 35105142.
- ^ a b Thomas Abrams (28 Baronial 2008). "Alert Letter" (PDF). Food and Drug Assistants.
FDA is non aware of whatever substantial evidence or substantial clinical experience that demonstrates that Bystolic represents a 'novel' or 'adjacent generation' beta blocker for the treatment of hypertension. Indeed, nosotros are not aware of any well-designed trials comparing Bystolic to other β-blockers. Furthermore, FDA is not aware of whatever data that would render Bystolic's mechanism of activity 'unique.'
[ expressionless link ]
External links [edit]
- "Nebivolol". Drug Data Portal. U.S. National Library of Medicine.
- "Nebivolol hydrochloride". Drug Data Portal. U.S. National Library of Medicine.
reinholdtanderies.blogspot.com
Source: https://en.wikipedia.org/wiki/Nebivolol
0 Response to "what blood pressure ill is equivalent to bystolic"
Postar um comentário